Side Effects With Inhaled Corticosteroids: Local Side Effects in Children
Similar findings for local side effects are seen for ICS/long-acting (3-agonist combination therapies. With BUD/formoterol, pharyngitis was seen in 6% of patients in clinical trials, with coughing in 5%. For FP/salmeterol, oropharyngeal candidiasis was seen in 2 to 4% of patients, hoarseness/dysphonia in 2 to 4%, throat irritation in 1 to 3%, and cough/breathing difficulties in 1 to 3%. In a comparative study of FP/salmeterol vs FP vs salmeterol vs placebo, there was no difference in the incidence of side effects between the ICS groups and the salmeterol group, except for a higher incidence of oral candidiasis for FP/salmeterol (10%) or FP alone (6%) vs salmeterol (3%) and placebo (< 1%).
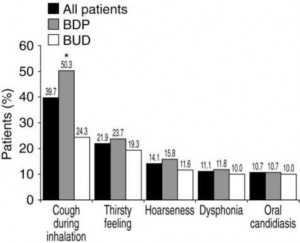
Studies on local side effects in children are less common than for adults. However, a relatively large, prospective, observational, cross-sectional cohort study of 639 patients (75.9 ± 48.9 months old) with moderate-to-severe asthma showed that 61.5% of patients had at least one local side effect (Fig 2). More info
The frequencies of side effects for BDP vs BUD are shown in Figure 2. There was no placebo control group. Cough during inhalation was significantly higher with BDP than with BUD; otherwise, the tolerability of the two therapies was similar. Dysphonia was the only side effect that appeared to be dose related, with a reduced frequency at low doses of BUD (p = 0.05). Perioral dermatitis and tongue hypertrophy were also observed in 2.9% and 0.1% of patients, respectively.
The investigators also studied the effect of device on local side effects. The main finding was that use of a spacer device doubled the incidence of cough during inhalation, and this was independent of the ICS used. These results were confirmed in a follow-up study in 219 patients, which noted cough on inhalation in 53.7% of patients using a spacer device; this was not linked to asthma severity, ICS, propellant or dispersant, or type of spacer, but was linked to therapy duration and use of long-acting (32-agonists. Dysphonia was more frequent with a pressurized metered-dose inhaler (pMDI) plus a spacer or a nebulizer vs the other devices. Perioral dermatitis was seen only in patients who used a spacer device with a facemask or a nebulizer with or without a facemask/mouthpiece.
Figure 2. Frequency of local side effects in asthmatic children for all patients and for those receiving either BDP or BUD. Dysphonia, hoarseness, cough, and thirst were obtained by patient questioning and oral candidiasis was defined as a clinical observation of thrush. Adapted from Dubus et al.7 *p = 0.0001 BDP vs BUD.