The general ED population

The most frequently referenced study supporting the efficacy of tadalafil is an integrated analysis of 5 randomized, double-blind, placebo controlled, multicentered phase III trials from 1112 men at 74 centers worldwide. The average age was 59, and the etiology of ED was 61% organic, 9% psychogenic, and 31% mixed. The ED severity at baseline was mild in 41%, moderate in 23%, and severe in 36%. Subjects were randomized to placebo or tadalafil at doses from 2.5 mg to 20 mg and instructed to self-administer a dose before initiating intercourse up to once daily. IIEF-EF, SEP, and GAQ scores were assessed at baseline and at 12 weeks.
Results of the integrated analysis found all doses of tadalafil to enhance efficacy outcomes. The IIEF-EF at the end of treatment increased by 6.5 in men receiving tadalafil 10 mg and by 7.9 with the 20 mg dose, compared with 0.6 in men receiving placebo (p < 0.001). Successful intercourse attempts were increased in 34% of men with a 10 mg dose and 39% with a 20 mg dose versus a 6% improvement in the placebo group when the SEP-Q3 answers were compared after 12 weeks (p < 0.001). Canadian Tadalafil remained efficacious up to 36 hours, as more than 70% of attempts were successful from 30 minutes to 36 hours after dosing. On the GAQ, 81% of men reported improved erections overall with tadalafil 20 mg compared with 35% in the placebo group (p < 0.001). The percentage of men receiving a 20 mg dose of tadalafil who had complete resolution of ED as defined by an IIEF-EF of 26 or higher was 59%, compared with only 11% with placebo.
In an update to Brock and colleagues’ study, Carson and associates performed another integrated analysis on all available tadalafil trials, including the original 1112 men in the 5 trials from Brock and colleagues’ original study, plus an additional 1215 men from 6 more recently completed trials. The same study design, safety measures, and statistical analysis were performed, except patients who received 2.5 mg and 5 mg doses in two of the earlier studies that were included in the analysis by Brock and colleagues were not included by Carson and associates. Only doses of 10 mg and 20 mg of tadalafil were compared with placebo. The average age was 56, and the etiology of ED was 58% organic, 12% psychogenic, and 31% mixed. The ED severity at baseline was mild in 36%, moderate in 27%, and severe in 33%. Comorbidities included 29% of men with hypertension, 16% with hyperlipidemia, 20% with diabetes mellitus, and 5% with coronary artery disease.
The results from the IIEF-EF, SEP-Q2, and SEP-Q3 are summarized in Figure 2. There were robust mean improvements in the IIEF-EF score, with increases of 6.5 points with 10 mg of tadalafil and 8.6 points with the 20 mg dose, versus only 0.9 points in the placebo group (p < 0.001). According to the SEP-Q3, successful intercourse attempts were improved by 34% to a total of 58% with 10 mg tadalafil, and a 46% improvement to a total of 68% was seen with the 20 mg dose, compared with an 8% improvement to a 31% total with placebo (p < 0.001). When successful intercourse attempts with tadalafil 20 mg were stratified by time after dosing, tadalafil remained almost equally efficacious at 36 hours. At the endpoint of the trial using the GAQ, 71% and 84% of patients reported overall improved erections with the two doses of tadalafil, respectively, compared with 33% with placebo (p < 0.001). This second integrated analysis of 11 tadalafil trials concluded that canadian tadalafil is a very effective and well tolerated treatment for ED of all severities and etiologies in men of all ages in the general population.
Figure 2. IIEF-EF, SEP-Q2, and SEP-Q3 improvements from baseline to 12 weeks with on-demand tadalafil (Cialis).
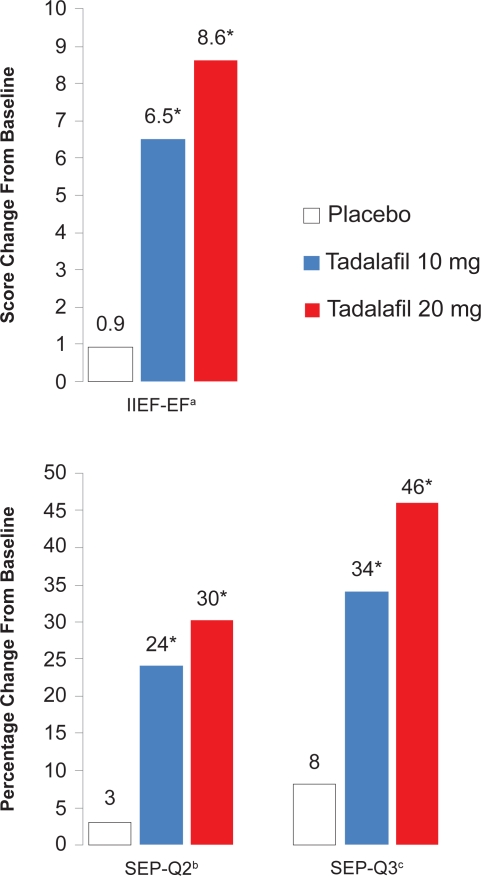
a-Erectile function domain score of the International Index of Erectile Function.
b-Question 2 from the Sexual Encounter Profile diary, “Were you able to insert your penis into your partner’s vagina?”
c-Question 3 from the Sexual Encounter Profile diary, “Did your erection last long enough for you to have successful intercourse?”
*-p< 0.001 versus placebo.
